Adicet Bio Announces Positive Preliminary Data from ADI-001 Phase 1 Study in Patients with Lupus Nephritis (LN) and Systemic Lupus Erythematosus (SLE)
-Clinical data to date supports a potentially transformational approach to treating autoimmune diseases with an off-the-shelf, one-time therapy-
-As of the August 31, 2025 data cut-off date, all seven evaluable LN and SLE patients experienced rapid and sustained reductions in the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI-2K) score and Physician's Global Assessment (PGA) highlighting ADI-001's potential for durable effect on a broad range of lupus symptoms-
-As of the August 31, 2025 data cut-off date, all five LN patients experienced improved renal function, including three complete renal responses and Definition Of Remission In Systemic lupus erythematosus (DORIS) remissions, and two partial renal responses, with all responses ongoing-
-Generally well tolerated with a favorable safety profile to date, supporting the potential for outpatient administration; no Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS) and no Grade 2 or higher Cytokine Release Syndrome (CRS) was observed-
-All patients discontinued immunosuppressants and either discontinued or tapered corticosteroids to physiological levels-
-Clear evidence of immune reset with subsequent emergence of naïve and previously undetected B cell repertoire following single treatment-
-Recent momentum has resulted in more than 25 clinical sites open for enrollment to date-
-Adicet plans to request a meeting with the U.S. Food and Drug Administration (FDA) in 1Q/2026 to inform Phase 2 pivotal trial design with a study anticipated to commence in 2Q/2026-
-Company to host investor webcast on Tuesday, October 7 at 8:00am ET-
Adicet Bio, Inc. (NASDAQ:ACET), a clinical stage biotechnology company discovering and developing allogeneic gamma delta T cell therapies for autoimmune diseases and cancer, today announced initial safety and efficacy data from the first seven patients dosed with ADI-001 in the ongoing Phase 1 study evaluating ADI-001 as a potential treatment for patients with autoimmune diseases. The data cut as of August 31, 2025, includes seven patients (five LN and two SLE patients), with follow-up ranging from two to nine months. Based on these results, Adicet will request a meeting with the FDA to inform trial design for a potentially pivotal Phase 2 trial for LN, or for LN and SLE.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20251006091236/en/
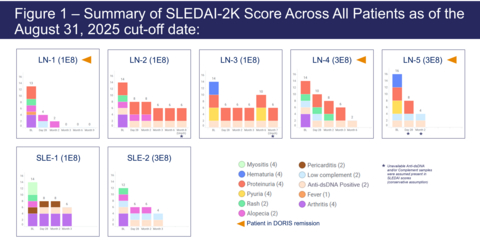
Figure 1 – Summary of SLEDAI-2K Score Across All Patients as of the August 31, 2025 cut-off date
"We are extremely encouraged to share preliminary results highlighting disease remissions or a halting of disease progression in the first seven patients treated in our Phase 1 trial. After a single dose of ADI-001, all patients discontinued immunosuppressant medications and all either discontinued or tapered corticosteroids to below physiological levels," said Julie Maltzman, M.D., Chief Medical Officer of Adicet Bio. "ADI-001 has also demonstrated a favorable safety and tolerability profile with no ICANS, and only two patients experiencing Grade 1 CRS, which is promising and has the potential to support outpatient administration of ADI-001. In addition, ADI-001's potential availability as an off-the-shelf therapy may allow for far broader accessibility to many more patients and treating physicians. We believe ADI-001 represents a potential paradigm shift in the treatment of autoimmune diseases that traditionally has required chronic therapy to treat unpredictable and dangerous flares, potentially resulting in end-organ damage."
Dr. Maltzman continued, "We have now activated over 25 clinical sites globally, which is a testament to our team's focus and execution as well as demonstrated investigator interest in ADI-001. The pace of enrollment and site activation we are now observing reinforces our confidence in our timelines for future milestones as we advance our Phase 1 study in LN and SLE as well as cohorts in other autoimmune diseases, including systemic sclerosis, idiopathic inflammatory myopathy, stiff person syndrome and anti-neutrophil cytoplasmic autoantibody associated vasculitis, as well as a new Phase 1 study that is now open for enrollment for the treatment of rheumatoid arthritis."
"These preliminary results reinforce our belief that ADI-001 has the potential to transform the treatment landscape for autoimmune diseases," said Chen Schor, President and Chief Executive Officer of Adicet Bio. "We observed clear evidence of immune reset with subsequent emergence of naïve B cell repertoire following a single treatment of ADI-001. ADI-001's potential to reset the immune system with a one-time, off-the-shelf therapy and a generally favorable safety profile could be practice-changing for patients and providers alike. With these data in hand, we plan to engage with the FDA in the first quarter of 2026 to discuss the design of a potentially pivotal study that we anticipate initiating in the second quarter of 2026."
Data highlights as of the August 31, 2025 cut-off date were as follows:
- Seven patients (five LN and two SLE) were evaluated with follow-up ranging from two to nine months.
- 100% of patients in the LN cohort achieved renal response, including three complete responses and DORIS remissions, and two partial responses, with all responses ongoing.
- 100% of patients saw rapid and sustained reductions in SLEDAI-2K and PGA scores, highlighting ADI-001's potential durable effect on a broad range of lupus symptoms.
- ADI-001 demonstrated multiple hallmarks of an immune reset with elimination of dominant B cell clones and subsequent emergence of naïve B cells and new B cell repertoire following single dose treatment.
-
As of August 31, 2025, ADI-001 was generally well tolerated and showed a favorable safety profile that could enable dosing in an outpatient setting.
- Across all seven patients dosed with ADI-001, there were no serious adverse events, and no reported cases of ICANS.
- Two patients experienced Grade 1 CRS (fever), and one patient had Grade 1 infection (respiratory tract infection).
- There were no cases of Graft-Versus-Host Disease (GvHD), Hemophagocytic Lymphohistiocytosis-Macrophage-Activation Syndrome (HLH-MAS), or prolonged neutropenia.
Anticipated development plans for ADI-001 are as follows:
- Adicet plans to request a meeting with the FDA in the first quarter of 2026 to inform Phase 2 pivotal trial design with a study anticipated to commence in the second quarter of 2026. SLE and LN patient enrollment to the ongoing Phase 1 is expected to continue until the Phase 2 pivotal study is open for enrollment. There are more than 25 clinical sites globally open for enrollment for the Phase 1 study of ADI-001 in autoimmune indications.
- The Phase 1 program is now open for enrollment of patients with systemic sclerosis (SSc), idiopathic inflammatory myopathy, stiff person syndrome, anti-neutrophil cytoplasmic autoantibody associated vasculitis and rheumatoid arthritis.
-
Below are the anticipated milestones for ADI-001 through the end of 2026:
-
First half of 2026:
- SLE and LN clinical update
- Alignment with FDA on pivotal study design in LN or LN/SLE
- Initiate pivotal study in LN or LN/SLE
- Clinical update in SSc
- Potential clinical update in other autoimmune indications
-
Second half of 2026:
- SLE and LN clinical update
- Clinical update in SSc
- Clinical update in RA with cyclophosphamide/fludarabine vs cyclophosphamide only conditioning
- Potential clinical update in other autoimmune indications
-
First half of 2026:
Table 1 – Summary of Phase 1 ADI-001 Preliminary Efficacy Data in LN Patients as of the August 31, 2025 cut-off date:
|
|
UPCR |
eGFR |
Renal response^ |
DORIS Remission |
||
Subj# |
Follow-Up (Months) |
Baseline |
Last Follow Up |
Baseline |
Last Follow Up |
||
LN-1 |
9 |
2.05 |
0.05 |
87 |
> 90 |
CRR at M1 |
DORIS at M6 |
LN-2 |
8 |
1.64 |
0.67 |
89 |
79 |
PRR at M8 |
|
LN-3 |
7 |
4.78 |
1.05 |
89 |
60 |
PRR at M7 |
|
LN-4 |
6 |
2.56 |
0.33 |
> 90 |
> 90 |
CRR at M5 |
DORIS at M5 |
LN-5 |
2 |
2.78 |
0.16 |
> 90 |
> 90 |
CRR at M2 |
DORIS at M2 |
^ Complete Renal Response (CRR)= UPCR ≤0.5 & EITHER eGFR ≥60 mL/min/1.73m2 OR no confirmed decrease from baseline in eGFR of >15% and no treatment or disease related eGFR-associated event
Partial Renal Response (PRR)= Reduction in baseline UPCR of ≥50% & final UPCR >0.5 to ≤3.0
DORIS Remission= Clinical SLEDAI (irrespective of serology)= 0 AND Physician global assessment score < 0.5; the subject may be on antimalarials, low-dose glucocorticoids (prednisolone ≤ 5 mg/day), and/or stable immunosuppressives including biologics.
DORIS= Definitions of remission in SLE initiative; eGFR= Estimated glomerular filtration rate; UPCR= Urine protein-to-creatinine ratio.
Table 2 – Summary of Phase 1 ADI-001 Safety Data in Efficacy Evaluable Patients as of the August 31, 2025 cut-off date:
|
1E8 |
3E8 |
All dose levels |
# of Patients |
4 |
3 |
7 |
SAEs |
-- |
-- |
-- |
CRS |
1 (Grade 1) |
1 (Grade 1) |
2 (28%) |
ICANS |
0 |
0 |
0 |
Infections |
1 (Grade 1) |
0 |
1 (14%) |
Safety assessment was performed using the Common Terminology Criteria for Adverse Events (v5) and the American Society for Transplantation and Cellular Therapy criteria (Lee 2018); CTCAE v5 Common Terminology Criteria for Adverse Events (CTCAE); Lee DW. Biol Blood Marrow Transplant. 2019 Apr;25(4):625-638.
Webcast/Conference Call Information
Adicet will host a webcast presentation on Tuesday, October 7 at 8:00am ET to discuss the most recent data, as of the August 31, 2025 data cut-off date, from its ongoing Phase 1 study evaluating the safety and tolerability of ADI-001 for the potential treatment of autoimmune diseases.
The live webcast of the presentation can be accessed by registering under "Presentations & Events" in the investors section of the Company's website at https://www.adicetbio.com. Upon registration, all participants will receive a confirmation email with a unique passcode to provide access to the webcast event. To participate via telephone, please join by dialing +1-646-876-9923 (domestic) or +44-330-088-5830 (international) and referencing the meeting ID 92057196728 and meeting passcode 958286. An archived replay will be available for 30 days following the presentation. The archived webcast will be available on the Company's website beginning approximately two hours after the event.
About ADI-001
ADI-001 is an investigational allogeneic gamma delta chimeric antigen receptor (CAR) T cell therapy targeting B-cells via an anti-CD20 CAR. ADI-001 was granted Fast Track Designation by the U.S. Food and Drug Administration (FDA) for the potential treatment of relapsed/refractory class III or class IV lupus nephritis (LN), refractory systemic lupus erythematosus (SLE) with extrarenal involvement, systemic sclerosis (SSc) and rheumatoid arthritis (RA).
About the Phase 1 Trial
The Phase 1 study has four separate arms, enrolling LN and SLE patients into one arm, SSc patients into a second arm, IIM, and SPS patients in a third arm and AAV patients into a fourth arm. Enrolled patients will receive a single dose of ADI-001. The dose-limiting toxicity window is 28 days with response and safety assessments conducted on Day 28 and during the follow up-period on months 3, 6, 9, 12, 18 and 24. The primary objectives of the study are to evaluate the safety and tolerability of ADI-001. Secondary objectives include measuring cellular kinetics, pharmacodynamics, changes in autoantibody titers, and appropriate disease activity scores in each indication.
For more information about becoming a study site, please email clinicaltrials@adicetbio.com or visit https://www.adicetbio.com/hcp/autoimmune/.
About Adicet Bio, Inc.
Adicet Bio, Inc. is a clinical stage biotechnology company discovering and developing allogeneic gamma delta T cell therapies for autoimmune diseases and cancer. Adicet is advancing a pipeline of "off-the-shelf" gamma delta T cells, engineered with chimeric antigen receptors (CARs), to facilitate durable activity in patients. For more information, please visit our website at https://www.adicetbio.com.
Forward-Looking Statements
This press release contains "forward-looking statements" of Adicet within the meaning of the Private Securities Litigation Reform Act of 1995 relating to the business and operations of Adicet. The words "anticipate," "believe," "continue," "could," "estimate," "expect," "intend," "may," "plan," "potential," "predict," "project," "should," "target," "would" and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements include, but are not limited to, express or implied statements regarding: clinical development of ADI-001, including the potential safety, tolerability and efficacy of ADI-001 for the treatment of autoimmune diseases; the expected progress, timing and success of the Phase 1 clinical trial of ADI-001 in autoimmune indications, including site activation, continued enrollment and expectations around the timing of future data releases; ADI-001's potential safety profile, availability as an off-the-shelf therapy and outpatient administration; expectations for and timing of future regulatory interactions, including alignment with the FDA for the potential initiation of a Phase 2 pivotal trial for ADI-001; and the potential for ADI-001 to be a paradigm shift in the treatment of autoimmune diseases.
Any forward-looking statements in this press release are based on management's current expectations and beliefs of future events, and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements, including without limitation, the effect of global economic conditions and public health emergencies on Adicet's business and financial results, including with respect to disruptions to our preclinical and clinical studies, business operations, employee hiring and retention, and ability to raise additional capital; Adicet's ability to execute on its strategy including obtaining the requisite regulatory approvals on the expected timeline, if at all; that positive results, including interim results, from a preclinical or clinical study may not necessarily be predictive of the results of future or ongoing studies; clinical studies may fail to demonstrate adequate safety and efficacy of Adicet's product candidates, which would prevent, delay, or limit the scope of regulatory approval and commercialization; and regulatory approval processes of the U.S. Food and Drug Administration and comparable foreign regulatory authorities are lengthy, time-consuming, and inherently unpredictable; and Adicet's ability to meet production and product release expectations. For a discussion of these and other risks and uncertainties, and other important factors, any of which could cause Adicet's actual results to differ from those contained in the forward-looking statements, see the section entitled "Risk Factors" in Adicet's most recent annual report on Form 10-K, quarterly reports on Form 10-Q and subsequent filings with the U.S. Securities and Exchange Commission (SEC), as well as discussions of potential risks, uncertainties, and other important factors in Adicet's other filings with the SEC. All information in this press release is as of the date of the release, and Adicet undertakes no duty to update this information unless required by law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20251006091236/en/
Adicet Bio, Inc.
Investor and Media Contacts
Anne Bowdidge
abowdidge@adicetbio.com
Penelope Belnap
Precision AQ
penelope.belnap@precisionaq.com
