Actinium Highlights Transcriptional Reprogramming as a Key Mechanism Underpinning Mutation-Agnostic Activity of Actimab-A in AML at the 2026 American Association of Cancer Research Annual Meeting
- Actimab-A combinations enhanced in vivo AML cell killing across multiple preclinical models, independent of mutation status, when combined with standard-of-care targeted and non-targeted therapies including revumenib (menin-KMT2A inhibitor), gilteritinib (FLT3 inhibitor), and azacitidine (hypomethylating agent) - three pillars of modern AML treatment - supporting its potential role as a universal combination backbone
- Transcriptional reprogramming identified as a central mechanism showing that Actimab-A combinations don't just add cytotoxicity, they reprogram AML cells from proliferation toward differentiation and apoptosis, providing the mechanistic basis for deeper, more durable MRD-negative responses and reinforcing Actimab-A's role as a universal combination backbone across AML
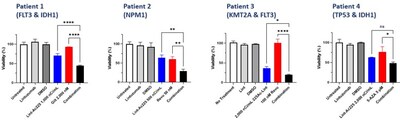
- Robust cytotoxicity observed in primary AML patient samples across key mutations (FLT3, KMT2A, NPM1, IDH1, IDH2, or TP53), reinforcing Actimab-A's potential as a mutation-agnostic backbone therapy, complementing the manageable safety profile of Actimab-A observed across prior clinical trials in over 150 AML patients
NEW YORK, April 22, 2026 /PRNewswire/ -- Actinium Pharmaceuticals, Inc. (NYSE AMERICAN: ATNM) (Actinium or the Company), a pioneer in the development of targeted radiotherapies, today highlighted data presented at the American Association for Cancer Research (AACR) Annual Meeting supporting transcriptional reprogramming as a central mechanism driving the mutation-agnostic anti-leukemic activity of Actimab-A (lintuzumab-Ac225) in acute myeloid leukemia (AML).
Preclinical translational data demonstrated that lintuzumab-Ac225 delivers potent cytotoxic activity across AML models harboring common mutations, including FLT3, NPM1, KMT2A, and TP53, as well as in primary patient samples. Importantly, combining Actimab-A with standard-of-care therapies - the menin inhibitor revumenib, the FLT3 inhibitor gilteritinib, and the hypomethylating agent azacitidine - resulted in enhanced leukemic cell killing in vivo across all tested models, independent of mutation status. These results support a combination-driven clinical strategy aimed at improving depth and durability of response. The findings provide the mechanistic foundation for Actimab-A's observed clinical activity and, together with the manageable safety profile demonstrated across prior Actimab-A trials in more than 150 AML patients, reinforce its suitability as a combination backbone across multiple treatment settings.
Actimab-A is Actinium's lead clinical radiotherapy delivering Actinium-225, a potent alpha-emitter radioisotope payload that produces lethal double-strand DNA breaks to kill CD33-expressing AML cells. CD33 is expressed ubiquitously in AML and other myeloid malignancies. Actimab-A has been evaluated in more than 150 AML patients across multiple treatment settings, including as monotherapy and in combination with the chemotherapy regimen CLAG-M and with the BCL-2 inhibitor venetoclax, where it has demonstrated compelling clinical activity and a manageable safety profile. Under our Cooperative Research and Development Agreement (CRADA) with the National Cancer Institute (NCI), Actimab-A is being advanced through the NCI's National Clinical Trials Network, including an ongoing frontline triplet trial combining Actimab-A with venetoclax and the hypomethylating agent ASTX-727 in newly diagnosed AML patients. The data presented at AACR 2026 further support Actimab-A's mutation-agnostic mechanism of action and its synergistic activity with targeted therapies approved for patients with the most commonly expressed AML mutations.
Key Data and Highlights of the Actimab-A AACR Presentation
New data on Actimab-A's mechanism and combination potential in primary AML patient samples further support its positioning as a foundational backbone therapy across multiple AML treatment settings, significantly expanding its commercial opportunity across the AML treatment continuum. In the relapsed/refractory AML setting Actimab-A in combination with the intensive chemotherapy regimen CLAG-M produced an 83% overall response rate and 75% MRD-negativity in a Phase 1 trial which forms the basis of a Phase 2/3 registrational study for which Actinium has FDA alignment and is seeking a development partner. Actimab-A is also being studied in newly diagnosed patients via the ongoing NCI-sponsored frontline triplet trial of Actimab-A with venetoclax and ASTX-727; and has shown promise in post-remission and MRD-directed settings; as well as myelodysplastic syndrome (MDS) and other CD33-expressing myeloid malignancies.
Transcriptional Reprogramming as a Key Mechanism for Actimab-A Combination Activity
- Combination treatment produced consistent pathway-level changes compared with monotherapy. Gene set enrichment analyses (GSEA) showed enhanced myeloid differentiation signatures with the addition of Actimab-A (lintuzumab-Ac225) to revumenib, gilteritinib, and azacitidine.
- Across models, combinations were associated with downregulation of proliferative programs, including MYC target genes, E2F targets, and G2/M checkpoint signatures, together with enrichment of p53-associated stress response and apoptosis pathways.
- Together, these findings show that Actimab-A combinations don't just add cytotoxicity — they reprogram AML cells from proliferation toward differentiation and apoptosis, providing the mechanistic basis for deeper, more durable MRD-negative responses and reinforcing Actimab-A's role as a universal combination backbone across AML.
Broad Activity of Actimab-A as Monotherapy and in Combination in Primary AML Patient Samples
- Actimab-A (LinT-Ac225) showed robust cytotoxicity in primary AML patient samples, independent of FLT3, KMT2A, NPM1, IDH1, IDH2, or TP53 mutation status, positioning Actimab-A to treat the full AML population, including TP53-mutant patients who lack effective targeted options, and to serve as a universal combination partner rather than a mutation-restricted therapy.
- Combining standard-of-care therapies (SOC) – revumenib (menin-KMT2A inhibitor), gilteritinib (FLT3 inhibitor), and azacitidine (hypomethylating agent) – with Actimab-A enhanced anti-leukemic efficacy across models - demonstrating synergy with one drug from each of the three pillars of modern AML care (targeted kinase inhibitors, menin inhibitors, and hypomethylating agents) and supporting Actimab-A's positioning as a universal combination partner across frontline, relapsed/refractory, and unfit AML populations.
Sandesh Seth, Actinium's Chairman and CEO, said, "The data presented at AACR 2026 represent a significant step forward in our mission to establish Actimab-A as the foundational backbone therapy across the AML treatment continuum. For the first time, we have clear molecular evidence – through transcriptional profiling – of how Actimab-A reprograms AML cells to activate p53-driven apoptosis and shut down proliferative signaling, providing the mechanistic basis for deeper, more durable MRD-negative responses and mutation-agnostic activity we have consistently observed clinically. As the only CD33-targeted radiotherapy in development for myeloid malignancies, Actimab-A uniquely leverages the broad, stable expression of CD33 and the potent, mutation-agnostic Ac-225 payload to complement – not compete with – the targeted therapies that define today's AML standard of care. Building on compelling clinical results across more than 150 patients in multiple treatment settings, and the high visibility of our NCI CRADA – including the ongoing frontline triplet trial with venetoclax and ASTX-727 – these findings will strengthen investigator enthusiasm for Actimab-A and reinforce the significant commercial opportunity ahead as we seek a partner for the registrational Phase 2/3 Actimab-A + CLAG-M study for which we have FDA alignment. We are focused on executing across our ongoing and planned clinical programs to deliver meaningful improvements in outcomes for AML patients, who continue to face high unmet medical need that is not addressed by currently available therapies."
The Actimab-A AACR presentation is available for viewing on the Presentations & Webinars page of Actinium's website HERE.
Title: Actimab-A, a CD33-Targeted Actinium-225 Radioconjugate, Drives Mutation-Agnostic Anti-Leukemic Activity and Synergizes with Standard Therapies in AML Through Transcriptional Reprogramming
Abstract Number: 5827
About Actimab-A
Actimab-A (lintuzumab-Ac225) is Actinium's lead CD33-targeted radiotherapy and the only CD33-targeted radiotherapy in clinical development for myeloid malignancies. Actimab-A pairs a humanized anti-CD33 monoclonal antibody (lintuzumab) with the potent alpha-emitter Actinium-225 (Ac-225), which delivers high-energy, short-range radiation that produces lethal double-strand DNA breaks in CD33-expressing leukemic cells while sparing surrounding healthy tissue. Because CD33 is expressed on the blasts of the large majority of AML patients and the Ac-225 payload kills cells independent of genetic background, Actimab-A is positioned as a mutation-agnostic backbone that can be combined with the targeted and non-targeted therapies that define today's AML standard of care.
Actimab-A has been studied in more than 150 patients with AML across multiple treatment settings, including as monotherapy and in combination with the intensive chemotherapy regimen CLAG-M and with the BCL-2 inhibitor venetoclax. In a Phase 1 trial in relapsed/refractory AML, Actimab-A plus CLAG-M produced an 83% overall response rate and 75% MRD-negativity at the recommended Phase 2 dose, with meaningful overall survival benefits in a high-risk population including patients with TP53 mutations and prior venetoclax exposure, and a manageable safety profile. Under a Cooperative Research and Development Agreement (CRADA) with the National Cancer Institute (NCI), Actimab-A is being advanced across the NCI's National Clinical Trials Network – which includes approximately 2,000 clinical trial sites through groups such as ECOG, SWOG, and Alliance – and is the subject of an ongoing frontline triplet combination trial with venetoclax and the hypomethylating agent ASTX-727 (Taiho Oncology) in newly diagnosed AML patients. Actimab-A is advancing toward a Phase 2/3 registrational program, with the goal of establishing Actimab-A as a foundational backbone therapy for patients with AML, myelodysplastic syndrome (MDS), and other CD33-expressing myeloid malignancies – a patient population that continues to face high unmet medical need.
About Actinium Pharmaceuticals, Inc.
Actinium is a pioneer in targeted radiotherapies designed to improve outcomes for patients with cancer. The company employs a biology-driven approach to develop differentiated radiopharmaceuticals for solid tumors and hematologic malignancies. Its mission is to transform cancer treatment through innovative radioconjugates that maximize therapeutic efficacy while minimizing toxicity to healthy tissue by combining expertise in tumor biology, translational medicine, and radiochemistry. Since inception, Actinium has focused on developing innovative radiotherapies. Its pipeline reflects this strategy across three areas: (1) solid tumor therapeutics including ATNM-400 and Actimab-A with pan-tumor potential; (2) Actimab-A as a therapeutic backbone for acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS) in collaboration with the National Cancer Institute (NCI); and (3) targeted conditioning agents including Iomab-B for bone marrow transplant and Iomab-ACT for cell and gene therapy conditioning. ATNM-400 targets a novel antigen distinct from PSMA and has demonstrated preclinical activity across metastatic castration-resistant prostate cancer (mCRPC), non-small cell lung cancer (NSCLC), and breast cancer. Actimab-A has shown improved survival in relapsed/refractory AML with CLAG-M and is advancing toward a Phase 2/3 trial, with additional development ongoing through a CRADA with the NCI. Actinium is also advancing preclinical solid tumor programs and holds ~250 patents and patent applications, including intellectual property related to cyclotron-based production of Ac-225. For more information, please visit www.actiniumpharma.com.
For more information, please visit: https://www.actiniumpharma.com/
Forward-Looking Statements
This press release may contain projections or other "forward-looking statements" within the meaning of the "safe-harbor" provisions of the private securities litigation reform act of 1995 regarding future events or the future financial performance of the Company which the Company undertakes no obligation to update. These statements are based on management's current expectations and are subject to risks and uncertainties that may cause actual results to differ materially from the anticipated or estimated future results, including the risks and uncertainties associated with preliminary study results varying from final results, estimates of potential markets for drugs under development, clinical trials, actions by the FDA and other governmental agencies, regulatory clearances, responses to regulatory matters, the market demand for and acceptance of Actinium's products and services, performance of clinical research organizations and other risks detailed from time to time in Actinium's filings with the Securities and Exchange Commission (the "SEC"), including without limitation its most recent annual report on form 10-K, subsequent quarterly reports on Forms 10-Q and Forms 8-K, each as amended and supplemented from time to time.
Investors:
[email protected]
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/actinium-highlights-transcriptional-reprogramming-as-a-key-mechanism-underpinning-mutation-agnostic-activity-of-actimab-a-in-aml-at-the-2026-american-association-of-cancer-research-annual-meeting-302749761.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/actinium-highlights-transcriptional-reprogramming-as-a-key-mechanism-underpinning-mutation-agnostic-activity-of-actimab-a-in-aml-at-the-2026-american-association-of-cancer-research-annual-meeting-302749761.html
SOURCE Actinium Pharmaceuticals, Inc.