Zimmer Biomet to Release New Data, Highlight Key Innovations at American Academy of Orthopaedic Surgeons Annual Meeting
Company's Chief Movement Officer, Arnold Schwarzenegger,
Will Be Presidential Guest Speaker
WARSAW, Ind., Feb. 25, 2026 /PRNewswire/ -- Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global medical technology leader, today announced it will unveil new clinical and operational data and showcase its newest innovations at the American Academy of Orthopaedic Surgeons (AAOS) 2026 Annual Meeting. The company's Chief Movement Officer Arnold Schwarzenegger will take center stage as the conference's Presidential Guest Speaker.
On Thursday, March 5 at 10:30 a.m. Central Time, Schwarzenegger will be featured as the Presidential Guest Speaker, and will join Zimmer Biomet Chairman, President and CEO Ivan Tornos and AAOS President Ned Amendola, MD, FAAOS, FRCSC, DABOS, for a main-stage conversation about sustaining peak performance, teamwork and thriving under pressure.
"Our presence at the AAOS Annual Meeting will highlight Zimmer Biomet's leadership in orthopedic implants coupled with the robotic and digital technologies that collect data before, during and after surgery to provide insights that help surgeons understand the potential impact of surgical decisions on patient outcomes," said Tornos. "In addition to the technologies we plan to showcase and new data being presented, we are very proud to feature our Chief Movement Officer, Arnold Schwarzenegger, as the Presidential Guest Speaker, and I look forward to joining him and Dr. Amendola for an engaging discussion."
The company will highlight its wide variety of orthopedic innovations and solutions at booth 4239, including its most differentiated technologies that have built a reputation as the "Magnificent Seven," including:
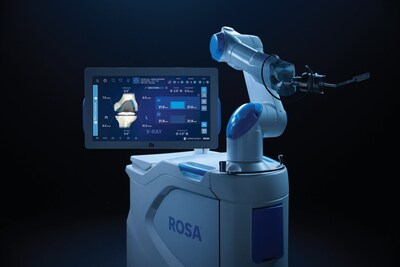
- Robotics and Navigation Technologies: From CT, to imageless to handheld robotics and AI-driven navigation technologies, Zimmer Biomet offers the industry's most comprehensive and adaptable suite of orthopedic robotics and navigation technologies designed to elevate surgical precision and transform patient outcomes.
- ROSA® Knee with OptimiZe™: The newest version of the ROSA® Knee System to customize and enhance the surgeon's experience with personalized and intelligent surgical planning, new positioning, tracking and alignment features to improve accuracy1 and reduce user variability2 — and offers the industry's only automated kinematic alignment feature to resurface the knee with the goal of restoring its pre-arthritic position and native joint lines.
- TMINI® Miniature Robotic System: A state-of-the-art miniature, handheld, wireless CT-based robotic system designed to enable accurate and precise3 implant placement.
- Monogram Technologies TKA System: A CT-based, semi-autonomous, total knee arthroplasty (TKA) robotic technology that received U.S. Food and Drug Administration (FDA) 510(k) clearance in March 2025. A surgeon-guided, fully autonomous version of this technology is currently in clinical trials.
- OrthoGrid Hip AI®: An AI-powered, fluoroscopy-based technology that provides hip surgeons with intuitive and instantaneous intra-operative tools to assist surgeons in achieving the desired surgical outcomes for component positioning.4
- Oxford® Cementless Partial Knee: Celebrating 50 years of clinical heritage, the Oxford Partial Knee is the most widely used5 and clinically proven6,7 partial knee system in the world. It is also the only FDA-approved mobile cementless partial knee implant in the U.S. that has been shown to be efficient8,9 in the operating room and has been proven to have excellent survivorship10,11 worldwide.
- Z1® Femoral Hip System: Offers an expansive size range and three distinct neck options designed to address a variety of patient anatomies and reconstructive needs.
- Persona IQ® The Smart Knee®: The first and only smart knee implant that captures patient-specific gait and range of motion metrics directly from the knee during patient monitoring to provide post-operative recovery insights12,13 and trends, allowing care teams to monitor and personalize the TKA patient experience.10-14
- Persona® OsseoTi® Keel Tibia: A cementless tibia for TKA with a 3D printed porous tray that provides stable initial and biological fixation and intra-operative versatility.15
- HAMMR® Automated Hip Impaction System: Designed to address surgeon strain, fatigue and repetitive motion associated with the traditional mallet during hip replacement procedures, and provides a consistent, controlled and precise energy16 designed to minimize variability in both bone preparation and device implantation.
- Persona® Revision Knee System: Expands the Persona Knee System to provide a portfolio of personalized knee care to replicate a patient's anatomy and offer an individualized fit in partial, primary and revision knee arthroplasties.
New data being presented at the conference will highlight:
- Safety and efficacy data for the Oxford® Cementless Partial Knee;
- Efficiency improvement metrics when implementing new processes and workflows in ambulatory surgery centers (ASCs);
- The relationship between early gait decline (collected with a smart implantable device) and periprosthetic joint infections in total knee arthroplasty (TKA);
- The relationship between qualified step counts (collected with a smart implantable device) and venous thromboembolisms (blood clots) following primary TKA;
- How the use of a smart implantable device and a digital care platform was associated with lower post-operative complications in knee replacement procedures;
- The association between 90-day readmissions and immediate post-operative qualified step counts (collected with a smart implantable device) following primary TKA; and
- New thresholds for post-operative gait metrics to help assess clinical significance in joint replacement patients.
Zimmer Biomet exhibit will also feature the latest innovations in shoulder reconstruction, sports medicine, trauma, foot and ankle reconstruction from the company's recent Paragon 28 acquisition, along with solutions for infection prevention and the company's ZBX™ Ambulatory Surgery Center Solutions (ASCs) — a comprehensive approach to help surgeons locate, design, build and operate ASCs.
About Zimmer Biomet
Zimmer Biomet is a global medical technology leader with a comprehensive portfolio designed to maximize mobility and improve health. We seamlessly transform the patient experience through our innovative products and suite of integrated digital and robotic technologies that leverage data, data analytics and artificial intelligence.
With 90+ years of trusted leadership and proven expertise, Zimmer Biomet is positioned to deliver the highest quality solutions to patients and providers. Our legacy continues to come to life today through our progressive culture of evolution and innovation.
For more information about our product portfolio, our operations in 25+ countries and sales in 100+ countries or about joining our team, visit www.zimmerbiomet.com or follow on LinkedIn at www.linkedin.com/company/zimmerbiomet or X at www.x.com/zimmerbiomet.
Persona IQ: The objective kinematic data generated by the CSE with CHIRP System are not intended to support clinical decision-making and have not been shown to provide any clinical benefit.
Cautionary Statement Regarding Forward-Looking Statements
This news release contains forward-looking statements within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include, but are not limited to, statements concerning Zimmer Biomet's expectations, plans, prospects, and product and service offerings, including new product launches and potential clinical successes. Such statements are based upon the current beliefs and expectations of management and are subject to significant risks, uncertainties and changes in circumstances that could cause actual outcomes and results to differ materially. For a list and description of some of such risks and uncertainties, see Zimmer Biomet's periodic reports filed with the U.S. Securities and Exchange Commission (SEC). These factors should not be construed as exhaustive and should be read in conjunction with the other cautionary statements that are included in Zimmer Biomet's filings with the SEC. Forward-looking statements speak only as of the date they are made, and Zimmer Biomet disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. Readers of this news release are cautioned not to rely on these forward-looking statements, since there can be no assurance that these forward-looking statements will prove to be accurate. This cautionary statement is applicable to all forward-looking statements contained in this news release.
Contacts: | |
Media | Investors |
Troy Kirkpatrick | David DeMartino |
614-284-1926 | 646-531-6115 |
Kirsten Fallon | Zach Weiner |
781-779-5561 | 908-591-6955 |
1 Data on File. DVaR-DS250106-01 ROSA Knee System v1.5 Validation Report.
2 Data on File. FER-EMS230714-01 Formative Evaluation Report - July Lab 2023.
3 Data on file at THINK Surgical. 2024. Data is based on TMINI System assisted placement of cut guide compared to planned placement.
4 Cardenas JM, Gordon D, Waddell BS, Kitziger KJ, Peters PC Jr, Gladnick BP. Does Artificial Intelligence Outperform Humans Using Fluoroscopic-Assisted Computer Navigation for Total Hip Arthroplasty? Arthroplasty Today. 2024 May 27;27:101410. doi: 10.1016/j.artd.2024.101410. PMID: 38840694; PMCID: PMC11150909.
5 Data on file at Zimmer Biomet. Based on Market Analysis and Registry Data, gathered September 2017.
6 Price, A. and Svard, U. A Second Decade Lifetable Survival Analysis of the Oxford Unicompartmental Knee Arthroplasty. Clin Orthop Relat Res. 2011 Jan;469(1): 174-9.
7 Pandit et al. The clinical outcome of minimally invasive phase 3 Oxford unicompartmental knee arthroplasty. Bone Joint J 2015;97-B:1493–1500.
8 Pandit, H., et al. "Improved fixation in cementless unicompartmental knee replacement: five-year results of a randomized controlled trial." JBJS 95.15 (2013): 1365-1372.
9 Stempin R, Kaczmarek W, Stempin K, Dutka J. Midterm Results of Cementless and Cemented Unicondylar Knee Arthroplasty with Mobile Meniscal Bearing: A Prospective Cohort Study. Open Orthop J. 2017 Oct 31;11:1173-1178. doi: 10.2174/1874325001711011173. PMID: 29290853; PMCID: PMC5721307.
10 NJR- UK . The National Joint Registry 22nd Annual Report 2025 London, 2024 [Available from: https://reports.njrcentre.org.uk/Portals/0/PDFdownloads/NJR%2022nd%20Annual%20Report%202025_Knees.pdf.
11 AOANJRR. Australian Orthopaedic Association National Joint Replacement Registry (AOANJRR). Hip, Knee & Shoulder Arthroplasty: 2023 Annual Report Adelaide, AOA2024 [updated 2024. Available from: https://aoanjrr.sahmri.com/documents/10180/1798900/AOANJRR+2024+Annual+Report.pdf/9d0bfe03-2282-8fc8-a424-b8d9abb82b1f?t=1727666185313.
12 Cushner FD, Yergler J, ElashoffB, Aubin PM, VertaP, Scuderi GR. Staying Ahead of the Curve: The Case for Recovery Curves in Total Knee Arthroplasty. The Journal of Arthroplasty. 2024;doi:10.1016/j.arth.2024.07.039
13 Cushner FD, Sculco PK, Long WJ. The Talking Knee Is a Reality: What Your Knee Can Tell You After Total Knee Arthroplasty. J OrthopaedicExperience and Innovation. 2022;2022
14 Cushner FD, Schiller P, Gross J, Mueller JK, Hunter W. A Total Knee Arthroplasty Prosthesis Capable of Remote Patient Monitoring. OrthopaedicProceedings. 2021/06/01 2021;103-B(SUPP_9):18-18 doi:10.1302/1358-992X.2021.9.018
15 Mueller J.K., et al. Persona OsseoTi Keel Tibia Provides Stable Initial Fixation 4027.2-GLBL-en. November 2022.
16 Data on File at Zimmer Biomet. Zimmer HAMMR Verification Test Report. DOC0179677. Pages 47-49. January 19, 2024.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/zimmer-biomet-to-release-new-data-highlight-key-innovations-at-american-academy-of-orthopaedic-surgeons-annual-meeting-302696434.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/zimmer-biomet-to-release-new-data-highlight-key-innovations-at-american-academy-of-orthopaedic-surgeons-annual-meeting-302696434.html
SOURCE Zimmer Biomet Holdings, Inc.