Pliant Therapeutics Announces Presentation of Updated Data from the Phase 1 Trial of PLN-101095 in Patients with ICI-Refractory Solid Tumors at the 2026 AACR Annual Meeting
Confirmed responses deepened with an average maximum tumor reduction from baseline of 89% and median time on treatment increased to 19 months
Data highlighted in oral presentation at AACR's Clinical Trials Mini Symposium
Phase 1b indication expansion trial is enrolling
SOUTH SAN FRANCISCO, Calif., April 18, 2026 (GLOBE NEWSWIRE) -- Pliant Therapeutics, Inc. (NASDAQ:PLRX) today announced the presentation of updated data from its Phase 1 trial of PLN-101095, in combination with pembrolizumab, in patients with immune checkpoint inhibitor (ICI)-refractory advanced or metastatic solid tumors. The oral presentation was made at the Clinical Trials Mini Symposium of the American Association for Cancer Research (AACR) 2026 Annual Meeting.
Timothy A. Yap, MBBS, Ph.D., Medical Oncologist and Physician-Scientist at the University of Texas MD Anderson Cancer Center, presented results from the dose escalation portion of the ongoing Phase 1a/1b trial covering data as of February 27, 2026. Results showed that in the heavily pretreated ICI-secondary refractory patient subgroup, twice daily (BID) treatment with PLN-101095 at the highest doses in combination with the ICI pembrolizumab showed antitumor activity. One confirmed overall complete response, two confirmed overall partial responses, including one patient with a complete response of baseline target lesions, and one unconfirmed partial response were reported in patients with cholangiocarcinoma, non-small cell lung cancer (NSCLC), melanoma and head and neck squamous cell carcinoma, respectively. As of the data cutoff date, the median time on treatment for the three confirmed responders was 19 months, who experienced an average baseline target tumor reduction of 89%.
All responding patients showed large increases (4- to 13-fold vs. baseline) in plasma interferon gamma (IFN-γ) after a 14-day run-in period of monotherapy with PLN-101095. At Week 10, all responders maintained more than a 2-fold increase in IFN-γ. No non-responders showed meaningful increases in IFN- γ. In addition to IFN- γ increases, all responding patients showed elevated plasma PD-L1 levels, known to be induced by increased IFN-γ and a predictor of an improved ICI response. As increases in IFN-γ have the potential to serve as a biomarker of TGF-β inhibition and an early indicator of PLN-101095 anti-tumor activity. Pliant anticipates further study of this biomarker as part of the expansion cohorts in the Phase 1b trial.
PLN-101095 was generally well tolerated across all doses evaluated with few discontinuations (n=2) due to adverse events. Rash was the most common treatment-related adverse event (TRAE), all Grade 1 or 2, and the majority of events occurred within the first 2 days of the initial pembrolizumab dose. One Grade 3 TRAE was observed.
PLN-101095 demonstrated a dose-ordered exposure at Day 14 with doses ≥1000 mg BID achieving sustained IC90 coverage and all participants dosed at ≥1000 mg BID maintaining IC75 coverage over 24 hours. These results support the consistent target engagement of PLN-101095.
Sixteen patients with ten different tumor types, including both primary and secondary refractory patients, were enrolled in five dose cohorts. Patients were treated for 14 days with PLN-101095 monotherapy administered orally at doses of 250 mg twice a day (BID) (n=1), 500 mg BID (n=2), 1000 mg BID (n=6), 1000 mg three times a day (TID) (n=4) or 2000 mg BID (n=3), followed by the addition of pembrolizumab at 200 mg administered intravenously every three weeks until disease progression. Scans were conducted at baseline, Day 14, Week 10, and every 8 weeks thereafter.
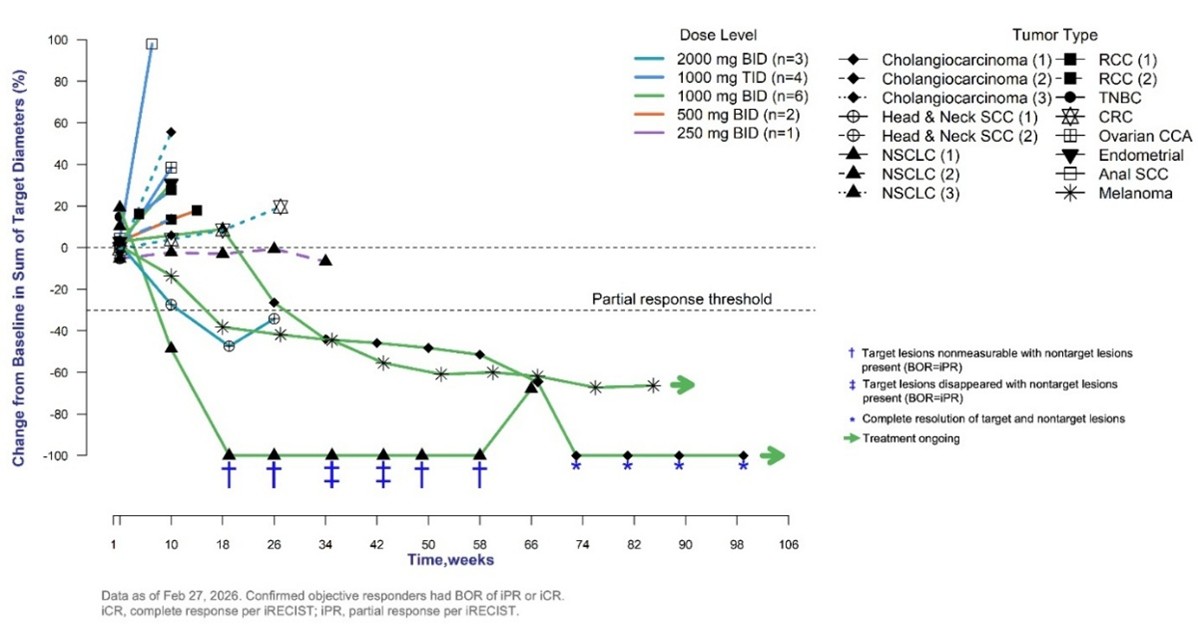
Figure 1. Percent Change from Baseline in Tumor Size by Week
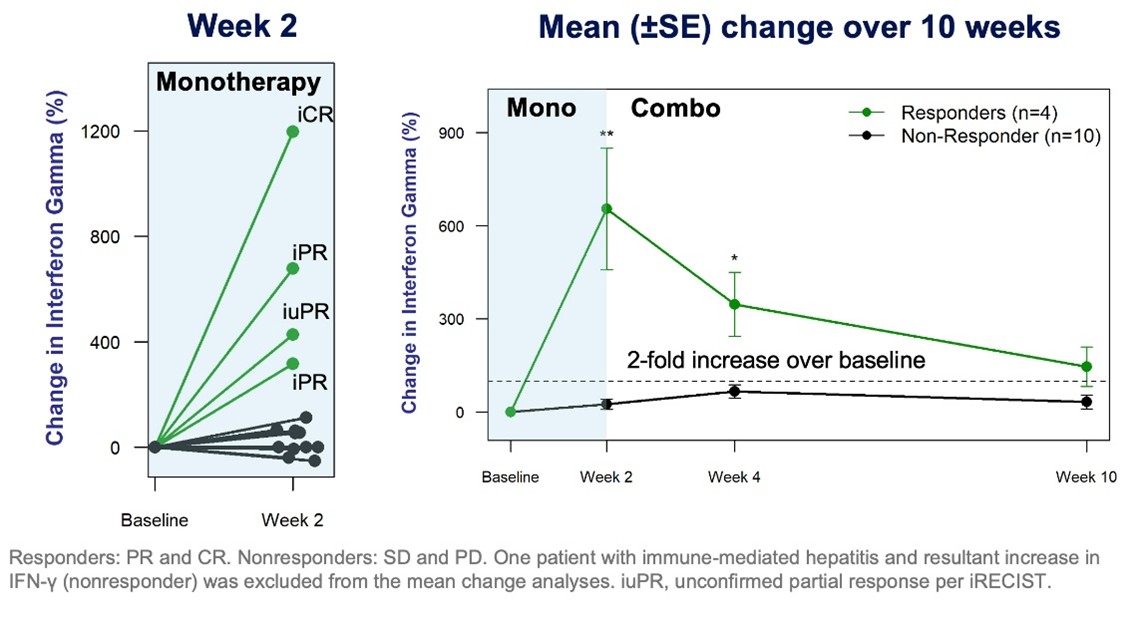
Figure 2. Percentage Change (+, -) from Baseline in Plasma IFN- γ
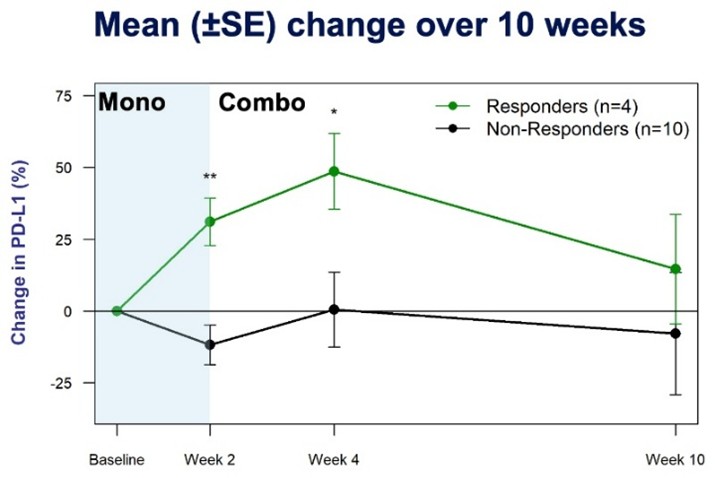
Figure 3. Percentage Change (+, -) from Baseline in PD-L1
Dr. Yap commented "One of the ways that the tumor microenvironment can suppress responses to immune checkpoint inhibitors is through a process that is activated by integrins to upregulate TGF-β. PLN-101095 is designed to inhibit the integrins before they can ever do that, which gives it significant potential to stimulate or reinvigorate the immune response to cancer. These clinical trial data, for the combination of PLN-101095 and pembrolizumab in patients with secondary immune checkpoint inhibitor resistance, show the potential to meet a high unmet therapeutic need."
"These encouraging results show a deepening of baseline tumor reductions in confirmed responders and an increased median time on treatment with PLN-101095 in patients with difficult-to-treat ICI refractory tumors," said Bernard Coulie, M.D., PhD., Chief Executive Officer of Pliant. "We have initiated the Phase 1b trial to expand this novel combination therapy in specific tumor types to address patients in need and deliver value to our investors."
Slides accompanying these data can be found here and under the Investors & Media page of Pliant's website at www.PliantRx.com.
PLN-101095 Phase 1b Indication Expansion Trial
Pliant has initiated a Phase 1b open-label indication expansion trial enrolling three cohorts of patients with NSCLC, tumors with high tumor mutational burden or clear cell renal cell carcinoma. Patients will be treated for 14 days with PLN-101095 dosed at 1,000 mg twice daily as monotherapy, after which pembrolizumab will be added as combination therapy. Enrollment is underway with interim data expected in 2027.
PLN-101095 for the Treatment of Checkpoint Resistant Tumors
PLN-101095 is an oral, small molecule inhibitor of integrins αvβ8 and αvβ1. PLN-101095 is being evaluated in Phase 1a/1b open-label, dose-escalation and indication expansion trial to evaluate the safety, tolerability, pharmacokinetics, and preliminary evidence of antitumor activity of PLN-101095 in combination with pembrolizumab, in patients with immune checkpoint inhibitor (ICI)-refractory advanced or metastatic solid tumors. Activated transforming growth factor-β (TGF-β) has been shown to foster an immuno-suppressive tumor microenvironment (TME) that contributes to immune-checkpoint inhibitor (ICI) resistance and treatment failure in cancer.1 Blocking integrins αvβ8 and αvβ1 has been shown to prevent the activation of TGF-β and is expected to stimulate immune activation by increasing immune cell infiltration into the tumor microenvironment.2,3 PLN-101095 in combination with an anti-PD-1 monoclonal antibody (mAb) elicited a dose-dependent reduction in tumor volume and increased CD8+ T cell tumor infiltration in the tumor microenvironment compared with anti-PD-1 mAb therapy alone.4 In preclinical studies, PLN-101095 demonstrated monotherapy activity in reduction of tumor volume and increased cluster of differentiation (CD)8+ T cell infiltration.
About Pliant Therapeutics, Inc.
Pliant Therapeutics is a clinical-stage biopharmaceutical company focused on the discovery and development of integrin-based therapeutics. Pliant's lead program is PLN-101095, a small molecule, dual-selective inhibitor of αvß8 and αvß1 integrins, that is being developed for the treatment of ICI-refractory advanced or metastatic solid tumors. PLN-101095 is being investigated in a Phase 1b indication expansion trial of PLN-101095 enrolling patients with NSCLC, tumors with high tumor mutational burden or clear cell renal cell carcinoma. Pliant's preclinical research is focused on tissue-specific delivery and internalization of drug payloads utilizing integrin receptor-binding molecules with programs focused on delivering siRNAs to skeletal muscle cells, adipocytes, and renal cells. For additional information, please visit: www.PliantRx.com. Follow Pliant on social media at X, LinkedIn and Facebook.
Forward-Looking Statements
Statements contained in this press release regarding matters that are not historical facts are "forward-looking statements" within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as "may," "will," "expect," "anticipate," "estimate," "intend," and similar expressions (as well as other words or expressions referencing future events, conditions, or circumstances) are intended to identify forward-looking statements. These statements include express or implied statements regarding ongoing clinical trial development of PLN-101095, including timing of enrollment and data; the potential benefits of PLN-101095; and Pliant's plans for the continued development of PLN-101095 among others, including its integrin-based drug delivery platform. Because such statements deal with future events and are based on Pliant's current expectations, they are subject to various risks and uncertainties and actual results, performance or achievements of Pliant could differ materially from those described in or implied by the statements in this press release. These forward-looking statements are subject to risks and uncertainties, including those related to the development and commercialization of Pliant's product candidates, including any delays in Pliant's ongoing or planned preclinical or clinical trials, the impact of current macroeconomic, geopolitical and marketplace conditions on Pliant's business, operations, clinical supply and plans, Pliant's reliance on single-source third parties located in foreign jurisdictions, including China, for critical aspects of Pliant development operations, the risks inherent in the drug development process, the risks regarding the accuracy of Pliant's estimates of expenses and timing of development, its capital requirements and the sufficiency of its cash to support Pliant's planned operations, and Pliant's ability to obtain and maintain intellectual property protection for Pliant's product candidates. These and additional risks are discussed in the sections titled "Risk Factors" and "Management's Discussion and Analysis of Financial Condition and Results of Operations" in Pliant's Annual Report on Form 10-K for the period ended December 31, 2025, and subsequent filings with the Securities and Exchange Commission (SEC), which are available on the SEC's website at www.sec.gov. Unless otherwise noted, Pliant is providing this information as of the date of this news release and does not undertake any obligation to update any forward-looking statements contained in this document as a result of new information, future events or otherwise.
Investor and Media Contact:
Christopher Keenan
Vice President, Investor Relations and Corporate Communications
Pliant Therapeutics, Inc.
ir@pliantrx.com
1 Pickup M. et al. Nat Rev Cancer. 2013 Nov;13(11):788-99.
2 Takasaka N. et al. JCI Insight. 2018 Oct 18;3(20).
3 Reed NI. et al. Sci Transl Med. 2015 May 20;7(288):288ra79.
4 Kothari V, et al. J Immunother Cancer. 2022;10(Suppl 2): A1403 abstract 1352 (SITC 2022)
Photos accompanying this announcement are available at:
https://www.globenewswire.com/NewsRoom/AttachmentNg/e31d4c37-738e-4662-861d-675c8c564317
https://www.globenewswire.com/NewsRoom/AttachmentNg/0a896cb8-2a3f-416a-8b87-de6e96b7349e
https://www.globenewswire.com/NewsRoom/AttachmentNg/cc7c4d1c-bbb5-4d9a-99d0-908629883b81


